
Salgenx LLC • Design • Develop • Analysis • Grid Scale Flow Battery
TEL: 1-608-238-6001 (Chicago Time Zone )
Email: greg@infinityturbine.com
3 MWh Grid Scale Saltwater Flow Battery that is Self Healing Consulting and Analysis Available More Info
Saltwater Flow Battery Technology Development Kit (TDK) Register your own IP with our package. Finish your own cathode then own your IP, then manufacture. Continue development of Volumetric Energy Density of 600 – 900 Wh/L. Buy sales leads, license or buy Salgenx name for your own production. More Info
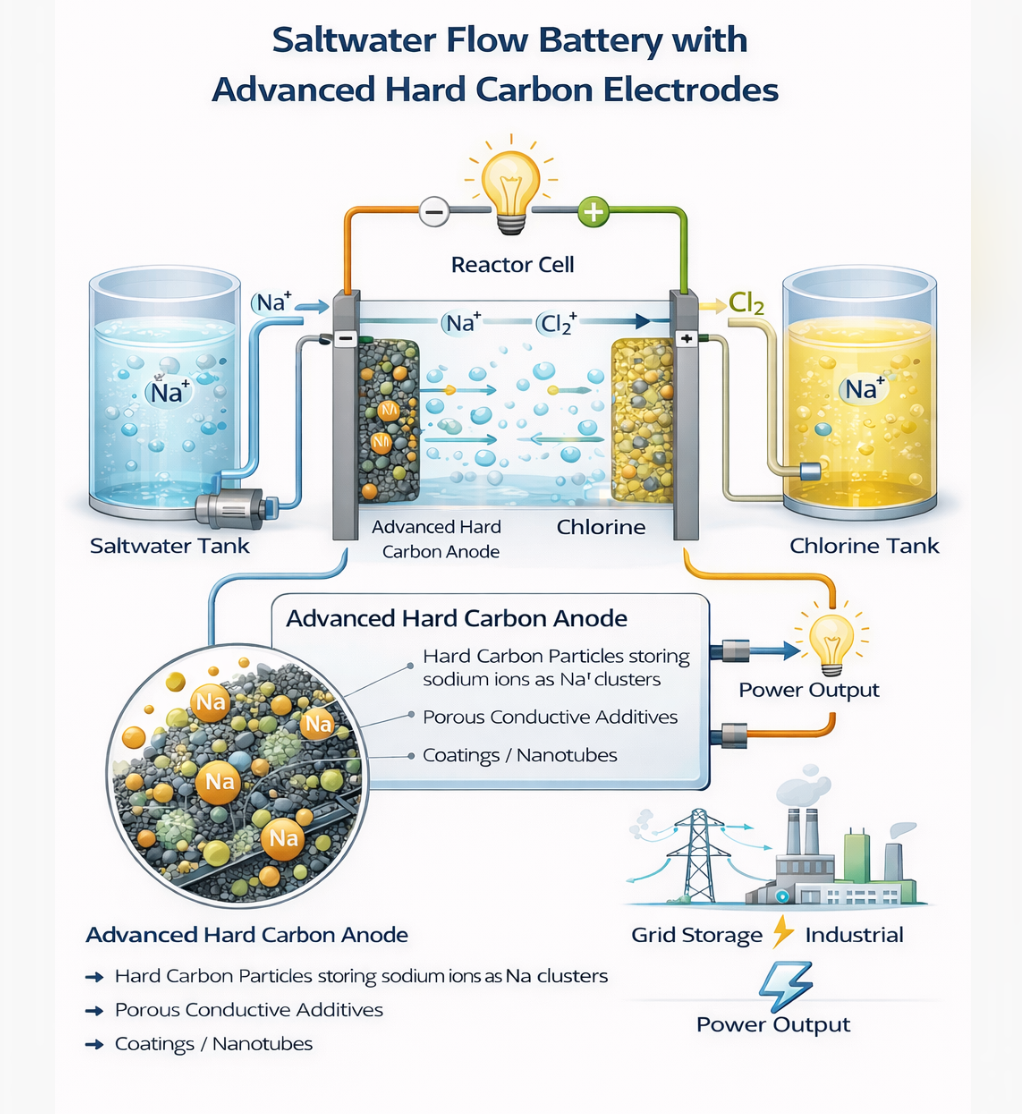
Salgenx is a modular saltwater electrochemical platform for producing grid-scale chemicals, hydrogen, and synthetic fuel feedstocks.
__________________________________________________________________________________________

|
|

|
|
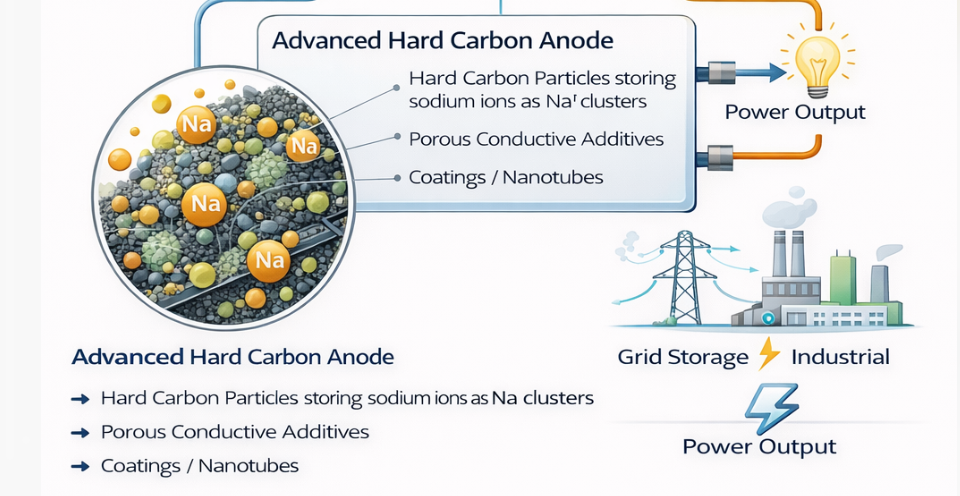
Breakthrough Findings from the Hard Carbon Kinetics Study Recent work using the diluted electrode method reveals that sodium insertion into hard carbon can exceed lithium in kinetic performance, with diffusion coefficients approaching 10 to the minus 10 square centimeters per second and lower activation energy. These findings provide a direct pathway to redesigning saltwater flow battery electrodes and enabling next generation ferro fluid sodium ion storage architectures.Hard carbon remains the leading anode candidate for sodium ion batteries and is increasingly relevant to hybrid aqueous and semi aqueous saltwater systems. The recent publication titled Revealing the kinetic limits of sodiation and lithiation at hard carbon using the diluted electrode method provides quantitative insight into the intrinsic reaction rates of sodium and lithium insertion in hard carbon, decoupled from composite electrode transport artifacts .This study is particularly important for flow battery developers because it isolates particle level kinetics from electrolyte depletion and porous electrode limitations. The conclusions materially affect how we should design electrodes in saltwater flow batteries, especially when considering ferro fluid style dispersed or slurry electrodes for sodium ion storage.Breakthrough Findings from the Hard Carbon Kinetics Study1. Sodium insertion is intrinsically faster than lithium in hard carbonUsing the diluted electrode method, the authors demonstrate that sodium insertion into hard carbon exhibits apparent diffusion coefficients on the order of 10 to the minus 10 to 10 to the minus 11 square centimeters per second, whereas lithium insertion ranges down to 10 to the minus 12 square centimeters per second .At slow rates involving both adsorption intercalation and pore filling, sodium diffusion was approximately one order of magnitude faster than lithium. This overturns the conventional assumption that lithium is always kinetically superior.2. Activation energy advantage for sodiumThe reported activation energies were approximately 55 kilojoules per mole for sodiation and 65 kilojoules per mole for lithiation .This lower energy barrier implies that sodium insertion into hard carbon becomes increasingly favorable at reduced temperatures, an important parameter for grid scale systems and outdoor stationary storage.3. Distinction between adsorption intercalation and pore filling kineticsThe study confirms two storage mechanisms:• Above 0.1 volts: adsorption and intercalation into pseudo graphitic domains• Below 0.1 volts: pore filling and pseudo metallic cluster formation At high charging rates, pore filling becomes kinetically limited and only the sloping adsorption region remains accessible.This is critical for flow batteries, where maintaining access to high capacity plateau regions under high current density is necessary for competitive power density.4. Charge transfer and solid state diffusion are dominant limitsThe diluted electrode method eliminated electrolyte depletion effects and showed that rate limitations are dominated by:• Charge transfer resistance at the electrolyte hard carbon interface• Solid state diffusion within hard carbon particles This means improving particle interface chemistry and reducing diffusion length are higher priority than simply increasing ionic conductivity in the bulk electrolyte.Application to a Saltwater Flow BatteryYour saltwater flow battery platform, especially with titanium ruthenium anode chemistry and either zinc or sodium titanium phosphate cathodes, can directly leverage these findings.1. Optimize hard carbon particle size and pore architectureBecause diffusion inside particles is limiting, decreasing particle radius significantly increases rate capability. The spherical diffusion model used in the paper shows capacity loss proportional to current and inversely proportional to diffusion coefficient and particle size .For a flow system, this suggests:• Use sub 5 micron hard carbon particles• Engineer uniform micropore size around 1 nanometer to favor sodium cluster formation• Increase graphitic domain connectivity without increasing excessive surface areaThe supporting information shows micropore size around 1.05 nanometers enabling pseudo metallic sodium clusters . This dimension should be a target design parameter for custom hard carbon synthesis in your system.2. Engineer a low resistance electrolyte interfaceSince charge transfer resistance dominates at high rate, surface functionalization becomes central. For aqueous saltwater systems:• Introduce oxygen and nitrogen functional groups to improve wettability• Apply thin conductive coatings such as carbon nanotube networks• Use electrocatalytically active interlayers to reduce desolvation barriersThe diluted electrode maintained conductivity above 100 siemens per meter even at 5 volume percent hard carbon using SWCNT networks .In your flow battery, a percolating conductive additive network inside the electrode tank or flow frame is essential to avoid rate collapse at high current density.3. Preserve access to pore filling at higher currentBecause plateau capacity is lost first at high rates, engineering strategies should focus on:• Increasing pore connectivity• Reducing tortuosity• Controlling electrolyte viscosityIn a saltwater flow system, this translates to lower viscosity electrolyte formulations and high flow shear conditions to maintain ion supply near particle surfaces.Implications for a Ferro Fluid Sodium Ion ElectrodeA ferro fluid electrode architecture introduces a fundamentally different geometry. Instead of fixed composite electrodes, you have mobile sodium storing particles suspended in a carrier liquid under magnetic control.The hard carbon kinetics study informs several design rules.1. Particle size becomes even more criticalBecause diffusion inside hard carbon particles limits performance, ferro fluid systems must:• Use ultrafine hard carbon particles• Maintain narrow particle size distribution• Prevent agglomerationThe supporting information shows particle D50 around 9 microns in the commercial sample . For ferro fluid use, that is too large. A target of 1 to 3 microns would reduce diffusion length by a factor of three to nine, dramatically increasing rate capability.2. Interface engineering replaces fixed SEI behaviorIn conventional lithium systems, SEI influences activation energy. For sodium ferro fluid systems in aqueous or hybrid electrolytes, a dynamic interphase will form.Given the lower activation energy of sodium insertion, maintaining a thin, stable interphase is essential to preserve this advantage. Strategies include:• Surface pre oxidation control• Use of inorganic passivation nanolayers• pH controlled electrolyte environments3. Magnetic structuring of conductive pathwaysSince the diluted electrode method shows charge transfer resistance is central, ferro fluid electrodes should not be randomly dispersed. Instead:• Use magnetic field alignment to form percolating chains• Create transient conductive scaffolds during charging• Collapse structure during idle for low sedimentationThis effectively recreates the conductive SWCNT network used in the diluted electrode but dynamically controlled.4. Exploiting sodium pseudo metallic cluster formationThe pore filling mechanism forms pseudo metallic sodium clusters below 0.1 volts .In a ferro fluid sodium storage system, this implies:• Hard carbon particles must contain controlled micropores• Pore entrances must not be blocked by surface coatings• Electrolyte desolvation energy must be minimizedIf successfully engineered, a ferro fluid sodium electrode could access both adsorption and pore filling capacity at high rates, potentially exceeding traditional fixed electrode sodium ion batteries in power density.Strategic Engineering ConclusionsThe diluted electrode study provides three decisive engineering directives:First, sodium kinetics in hard carbon are fundamentally competitive and in some regimes superior to lithium.Second, particle level diffusion and interface charge transfer dominate performance once electrolyte transport is resolved.Third, pore filling kinetics must be specifically engineered if high power density and high energy density are both desired.For your saltwater flow battery platform and ferro fluid sodium ion electrode concept, this translates to:• Custom synthesized low surface area, controlled micropore hard carbon• Sub micron to few micron particle sizing• Aggressive interface engineering• Dynamic conductive network formation in slurry systems• Electrolyte formulations optimized for rapid sodium desolvationIf implemented coherently, these advances can shift sodium based flow systems from low power grid storage to high power industrial or AI data center buffering applications. |
|
INVESTOR TECHNICAL SUMMARY Hard Carbon Sodium Storage Breakthrough and Impact on Saltwater Flow and Ferro Fluid Battery PlatformsCORE SCIENTIFIC BREAKTHROUGH• Sodium insertion into hard carbon is intrinsically faster than lithium in key operating regimes• Sodium diffusion coefficients up to approximately 10^-10 cm^2 per second• Activation energy lower for sodium than lithium• Two storage mechanisms confirmed– Surface adsorption and intercalation– Micropore filling forming pseudo metallic sodium clusters• Performance limits are dominated by particle level diffusion and interface charge transfer rather than bulk electrolyte transportWHY THIS MATTERS FOR GRID SCALE STORAGE• Faster sodium kinetics enable higher charge and discharge rates• Lower activation energy improves performance at lower temperatures• Hard carbon can deliver lithium comparable energy density with lower cost materials• Kinetic limits now clearly defined enabling targeted engineering optimizationDIRECT ADVANTAGES FOR SALTWATER FLOW BATTERIES• Enables high power density sodium storage using inexpensive carbon materials• Flow systems can eliminate electrolyte depletion limitations identified in traditional batteries• Particle engineering can unlock full plateau capacity at higher current density• Conductive additive networks dramatically improve charge transfer rates• System level optimization possible through particle size, pore structure, and flow dynamicsENGINEERING DESIGN TARGETS FOR NEXT GENERATION ELECTRODES• Hard carbon particle size preferably below 5 microns• Controlled micropore diameter near 1 nanometer• Low surface area to reduce irreversible capacity• High conductivity percolation network within electrode structure• Surface chemistry optimized for rapid sodium desolvation and insertionFERRO FLUID SODIUM ELECTRODE OPPORTUNITY• Mobile sodium storing particles suspended in conductive fluid• Magnetic alignment can create dynamic conductive pathways• Continuous flow improves ion supply to particle surfaces• Eliminates fixed electrode transport limitations• Enables high surface area active material with controlled diffusion distanceEXPECTED PERFORMANCE BENEFITS• Higher charge rate capability than conventional sodium ion batteries• Improved power density for industrial and data center buffering• Enhanced cold temperature performance• Scalable architecture compatible with containerized storage systems• Lower materials cost relative to lithium based systemsCOMMERCIAL IMPACT• Strengthens economic case for sodium based grid storage• Supports high cycle life long duration storage platforms• Enables high power hybrid energy buffering applications• Provides technical pathway to slurry based and flow based sodium batteries• Creates foundation for ferro fluid electrochemical storage systemsSTRATEGIC POSITIONING• Technology aligns with low cost abundant sodium supply chain• Compatible with modular containerized battery manufacturing• Supports large scale renewable integration• Enables differentiated intellectual property around dynamic electrodes and flow architecturesINVESTMENT THESIS• Clear kinetic advantage of sodium in engineered hard carbon• Identified and solvable rate limiting mechanisms• Multiple scalable implementation pathways– Fixed electrode saltwater flow batteries– Slurry electrode systems– Magnetically structured ferro fluid storage• High probability of performance gains through materials engineering rather than fundamental chemistry riskBOTTOM LINESodium hard carbon storage is no longer limited by unknown reaction behavior. The kinetics are quantified, the limiting mechanisms are identified, and engineering pathways exist to deliver higher power, lower cost, and scalable energy storage systems suitable for grid and industrial deployment. |
| CONTACT TEL: +1 608-238-6001 (Chicago Time Zone) Email: greg@infinityturbine.com | AMP | PDF | Salgenx is a division of Infinity Turbine LLC |